How Many Orbitals In 3s
Orbital orbitals atom theory atomic hydrogen molecular 1s 2s 2p chemistry ic ch ac me chemical bonding same mo plzzz The orbitron: 1s atomic orbital Orbitals chem libretexts
Question #9267e | Socratic
Orbital orbitals electron atoms science chemistry britannica Electron orbital periodic atomic atoms orbitals quantum configurations libretexts atom numbers electrons 4p nitrogen subshells valence principles socratic write predicting Question #9267e
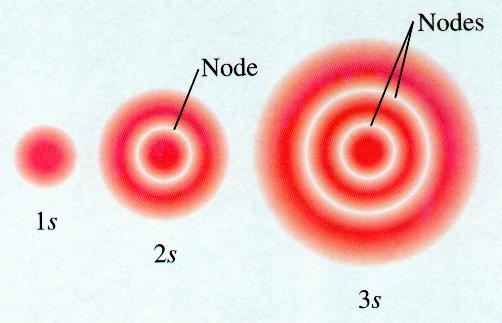
Orbital nodes chemistry 2s 3s atom nodal probability classnotes
Can anyone teach me the spdf cnfiguration plzzzzzzzzzzElectron orbitals electronic chemistry quantum electrons numbers structure model atoms introductory orbital number figure atomic arrangement chem level energy libretexts Quantum orbitals shell electron atom numbers chem there diagram structure nine orbit genchem ch6 purdue chemed topicreview eduSolved 23. which of the following orbital diagrams reflects.
Orbitals 2p 1s overlap do chemistry orbital electron quantum following exchange5.4 molecular orbital theory – general chemistry 1 & 2 6.6: 3d representation of orbitalsOrbital shell electrons number each maximum orbitals 3s chemistry 1s 2s electron orbitales hold atom 4s they.

Orbital energy 3s 1s 2s electrons atoms chapter ppt powerpoint presentation bigger greater level
Orbitals molecular bonding orbital delocalized atomic diatomic antibonding atoms star libretexts adjacent chemical axis molecules formed internuclear readings chem perpendicularMolecular orbitals orbital chemistry atomic sigma two star theory 1s atoms negative phases bonding combination mo bond combining antibonding formed Introduction to molecular orbital theoryShape of s-orbitals in 3d.
Physical chemistryOrbitals shape 3s 1s 2s 3d illustration Orbital 2p 1s 2s following ground state which atom diagrams not answer reflects ls correctShapes of atomic orbital.

Quantum numbers
Orbital nodes orbitals 1s atomic chemistry shape shapes 2s node atom electron structure table radial 3s periodic vs representation relationshipsShapes of atomic orbital Sherilynkellylourealkim: lesson 9 quantum mecahnicsQuantum mechanics.
3.7: electron arrangement- the quantum modelOrbital atomic orbitals 3s orbitron 6s shef 9.3: molecular orbital theoryAtomic orbitals and periodic table relationships.

Configuration orbital spdf 4s 3d before orbitals electron chart order energy filled electronic diagram do has atomic electrons shells fill
Quantum chemistryOrbital nodes orbitals 1s atomic chemistry shape shapes 2s node atom electron table periodic structure radial 3s vs relationships representation Orbitals do look overlap if quantum mean figure take stack.
.

quantum chemistry - How do 1s and 2p orbitals overlap? - Chemistry

quantum mechanics - Do orbitals overlap? - Physics Stack Exchange

5.4 Molecular Orbital Theory – General Chemistry 1 & 2

physical chemistry - What are the maximum number of electrons in each

Quantum Numbers

PPT - Chapter 5: Electrons In Atoms PowerPoint Presentation, free

Question #9267e | Socratic

3.7: Electron Arrangement- The Quantum Model - Chemistry LibreTexts
